DynaMesh-IPST
$0.00
Shipped From Abroad
DynaMesh®-IPST, DynaMesh®-IPST visible, DynaMesh®-IPST-D visible, DynaMesh®-IPST-R and DynaMesh®-IPST-R visible implants are intended for the surgical treatment of parastomal hernias, and the prevention (not DynaMesh®-IPST-R or DynaMesh®-IPST-R visible) of parastomal hernias following ostomy surgery, and permanently bridge and reinforce the soft tissue of the abdominal wall in the area of the stoma.
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
Description

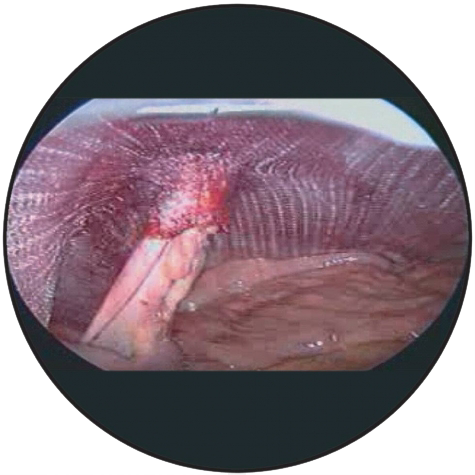
3D Funnel
The device has a passage point with a connected funnel for the passage of the intestine. The base area of the device must be positioned in contact with the tissue, as flat and smooth as possible, with the funnel pointing in the direction of the abdominal cavity.
When selecting the device, the diameter of the passage point should be chosen so that the intestine is tightly sheathed in the funnel.

Prefabricated Slit & Smooth Warp-Knitted Selvedges
Only DynaMesh®-IPST-R and DynaMesh®-IPST-R visible have a one-sided slit along the funnel and the base area (mesh flaps), starting from the passage point.
During positioning, the tight sheathing of the intestine in the funnel is achieved through appropriate overlapping of the mesh flaps with non-absorbable closure of the aperture.
Specifications
| Product | DynaMesh®-IPST (1) DynaMesh®-IPST visible (2) DynaMesh®-IPST-D visible (3) DynaMesh®-IPST-R (4) DynaMesh®-IPST-R visible (5) |
| Surgical Treatment | Parastomal Hernia (Repair / Prevention) (1) – (3) / (Repair) (4) (5) |
| Surgical Approach | Minimally Invasive / Open |
| Surgical Technique | Chimney Technique |
| Mesh Position | Intraperitoneal |
| Fixation | Suture / Tacks / No Fibrin Glue |
| Green Filament Ends |
|
| Smooth Warp-Knitted Selvedges | |
| Visible Technology |
|
| Materials | – Polyvinylidene fluoride (PVDF) (CAS 24937-79-9) > 85% (w/w) (1) – (5) – Polypropylene (PP) (CAS 9003-07-0) < 13% (w/w) (1) – (5) – Phthalocyanine green (CAS 1328-53-6) < 1% (w/w) (1) – (5) – Triiron tetraoxide (CAS 1317-61-9) < 1% (w/w) (2) (3) (5) |
| Polymers (Monofilament) | PVDF, PP |
| Biocompatibility |
|
| Ageing Resistance |
|
| Effective Porosity |
|
| Klinge’s Mesh Classification | Class 1a [102P, TR71B] |
| Applies to all product sizes | |
| Does not apply | |
| [#] | Reference “#” (see “References”) |
| [TR#] | Internal test report (see “internal test report references”) |
| Limitations | “A” animal trial, “B” bench test, “VIT” in-vitro trial, “P” published results based on the analysis of human mesh explants, “PB” published results mainly based on bench tests |
DynaMesh®-IPST implants (1)-(5) must be inserted via an appropriate minimally invasive or open approach and must be placed intraperitoneally.
DynaMesh®-IPST implants (1)-(5) have a parietal side and a visceral side. The parietal side is identified by green filament ends and consists of PVDF on the surface and a small proportion of PP, whereas the visceral side consists of PVDF on the surface.
When using DynaMesh®-IPST-D visible (3), attention must be paid to the side specificity (left-sided/right-sided stoma), through which a particularly large overlap in a cranial as well as in a medial direction is ensured.
When positioning DynaMesh®-IPST-R and DynaMesh®-IPST-R visible (4) (5), the tight sheathing of the intestine in the funnel is achieved through appropriate overlapping of the mesh flaps with non-absorbable closure of the aperture
Quick Comparison
| DynaMesh-IPST remove | Electric Suction Machine remove | 18Ltrs Bucket Autoclave remove | Ambu Oval Silicone resuscitator (Ambu Bag) remove | Oxygen Regulator (Japan Technology) remove | IBIS Neeo R9 Digital Surgical C-Arm remove | |||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | DynaMesh-IPST remove | Electric Suction Machine remove | 18Ltrs Bucket Autoclave remove | Ambu Oval Silicone resuscitator (Ambu Bag) remove | Oxygen Regulator (Japan Technology) remove | IBIS Neeo R9 Digital Surgical C-Arm remove | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image |  |  |  |  |  |  | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SKU | SF1033560130161-6 | SF1033560084-16 | SF1033560084-7 | SF1033560084-62 | SF1033560084-48 | SF1033560011-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rating | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Price |
| $152.00 |
| $27.00 | $17.00 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock | In stock | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Availability | In stock | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Add to cart | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Shipped From Abroad
DynaMesh®-IPST, DynaMesh®-IPST visible, DynaMesh®-IPST-D visible, DynaMesh®-IPST-R and DynaMesh®-IPST-R visible implants are intended for the surgical treatment of parastomal hernias, and the prevention (not DynaMesh®-IPST-R or DynaMesh®-IPST-R visible) of parastomal hernias following ostomy surgery, and permanently bridge and reinforce the soft tissue of the abdominal wall in the area of the stoma.
Delivery & Availability:
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
| In Stock
Power consumption: 120W
Maximum negative pressure: ≥0.09 MPa
Pump rate: ≥20 L/min
Noise: ≤60 dB
Continuous Working Time: 0-30 min
Bottle volume: 2500 ml, 2 pcs
Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In Stock
| In Stock The Ambu Oval Silicone resuscitator is designed for manual ventilation of neonates though to adults. Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In stock
| Shipped from Abroad Our Neeo “C” arms are easy to place, use and are specifically designed to be used in orthopedics, traumatology, abdominal surgery, urology, cardiology and operating rooms. Delivery & Availability: Typically 21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Content |
3D Funnel
The device has a passage point with a connected funnel for the passage of the intestine. The base area of the device must be positioned in contact with the tissue, as flat and smooth as possible, with the funnel pointing in the direction of the abdominal cavity.
When selecting the device, the diameter of the passage point should be chosen so that the intestine is tightly sheathed in the funnel.
Prefabricated Slit & Smooth Warp-Knitted Selvedges Only DynaMesh®-IPST-R and DynaMesh®-IPST-R visible have a one-sided slit along the funnel and the base area (mesh flaps), starting from the passage point. During positioning, the tight sheathing of the intestine in the funnel is achieved through appropriate overlapping of the mesh flaps with non-absorbable closure of the aperture. Specifications
| Specification :
Voltage: AC 220V, 50Hz or 110V, 60Hz
Power consumption: 120 W
Maximum negative pressure: ≥0.09 MPa
Negative pressure range: 0.02 MPa to Max negative pressure
Pump rate: ≥20 L/min
Noise: ≤60 dB
Continuous Working Time: 0-30 min
Rest Time≥30min
Bottle volume: 2500 ml, 2 pcs
Accessory: pedal switch (1pc), power switch(1pc),
filter(2 pcs), suction pipe(1pc, 2m in length, silica gel),
celiac suction pipe(1pc, 31cm in length, chrome copper)
Package: 1pc/carton
Box Size : 46.5*41.5*64.5cm
Net Weight: 11.5KGS
Gross Weight:13.8 KGS
Click Here To Download Catalogue | Features:
| The Ambu Oval Silicone resuscitator is designed for manual ventilation of neonates though to adults.
Features
* International guidelines recommend that all manual resuscitators for infant and pediatric patients should be equipped with a pressure limitation mechanism so the airway pressure does not exceed 45 cm H2O. |
| Our Neeo “C” arms are easy to place, use and are specifically designed to be used in orthopedics, traumatology, abdominal surgery, urology, cardiology and operating rooms.
Using Neeo with the RTP (Real Time Processing) option it is possible to perform vascular, urological and cardiological diagnostics. One of the main functions, digital image subtraction, allows to see, as an example, the passage of contrast liquids in a tissue or in a venous or arterial duct; thanks to the possibility of looping, the acquired video can be reproduced several times to monitor more accurately the passage of the fluid within the area in question. Angiographic measurement is another useful function in the vascular field (QA Quantitative Angiography) that allows the measurement of stenoses. Finally, fluoroscopy allows the correct positioning of stents or expanders.
Neeo is used in various interventional and diagnostic procedures in traumatology and orthopedics wards and operating rooms as well. Thanks to low-dose fluoroscopy, it is possible to use the device for positioning bone or subcutaneous grafts, inserting K-wire (Kirschner wire) for stabilization of bone fragments or for the correct positioning of prostheses. The low dose emitted ensures safe use for both the patient and the surgeon or doctor on the operating field.
On the control panel there is a large touch screen display that allows to adjust the basic functions of the equipment. From this display it is possible to select and adjust the fluoroscopic data for the examination, activate or deactivate the laser pointer, select between pulsed, one shot or standard fluoroscopy, rotate the image and perform all operations on collimator. The four side buttons on the display offer the possibility to move the bow vertically thanks to an extremely silent motor.
Neeo has two 19 “medical grade monitors that can be positioned according to the needs of the medical practitioner. Work monitors and feedback monitors are separated to be managed independently. The possible movements are: rotation, revolution, tilting and possibility of height adjustment.
Features:
Click Here To Download Catalogue | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dimensions | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additional information |







Reviews
There are no reviews yet.