DynaMesh-ENDOLAP
$0.00
Shipped From Abroad
DynaMesh-ENDOLAP and DynaMesh-ENDOLAP visible implants are intended for the surgical treatment of inguinal or femoral hernias and permanently bridge and reinforce the soft tissue in the groin region in the area of the hernia defect.
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
Description

Smooth Warp-Knitted Selvedges
The devices have smooth warp-knitted selvedges.

Choice of Method
The devices must be placed by means of laparoscopic transabdominal preperitoneal (TAPP) or endoscopic totally extraperitoneal (TEP)1) hernia repair. The colouring provides better intraoperative visibility and handling of the device.
1) Image of surgery courtesy of Dr. A. Kuthe,
DRK-Krankenhaus Clementinenhaus, Hanover, Germany
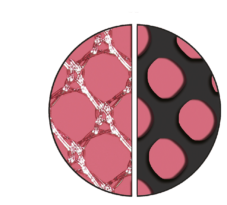
High Effective Porosity
The devices have a high effective porosity, which reduces inflammation and the risk of excessive scar formation. [103P, TR23]
Product Range
When selecting and cutting the device, sufficient defect overlap must be ensured to minimise the likelihood of the hernia recurring.
| DynaMesh®-ENDOLAP | 7.5 cm x 15 cm | PV100715F1/F3 |
| DynaMesh®-ENDOLAP | 10 cm x 15 cm | PV101015F1/F3/F10 |
| DynaMesh®-ENDOLAP | 12 cm x 15 cm | PV101215F1/F3/F10 |
| DynaMesh®-ENDOLAP | 13 cm x 15 cm | PV101315F1/F3 |
| DynaMesh®-ENDOLAP | 13 cm x 17 cm | PV101317F1/F3/F10 |
| DynaMesh®-ENDOLAP | 15 cm x 15 cm | PV101515F1/F3/F10 |
| DynaMesh®-ENDOLAP visible | 10 cm x 15 cm | PV141015F1/F3/F10 |
| DynaMesh®-ENDOLAP visible | 15 cm x 15 cm | PV141515F1/F3/F10 |
| DynaMesh®-ENDOLAP visible | 15 cm x 30 cm | PV141530F1 |
| FX = X unit(s)/box (e.g. F3 = 3 unit(s)/box) |
Specifications
| Product | DynaMesh®-ENDOLAP (1) DynaMesh®-ENDOLAP visible (2) |
| Surgical Treatment | Inguinal Hernias / Femoral Hernias |
| Surgical Approach | Laparoscopic / Endoscopic |
| Surgical Technique | TAPP / TEP |
| Mesh Position | Preperitoneal (Posterior) |
| Fixation | (None)* / Sutures / Tissue Adhesives / Tacks |
| Coloured Filaments | Green |
| Smooth Warp-Knitted Selvedges |
|
| Visible Technology |
|
| Materials | – Polyvinylidene fluoride (PVDF) (CAS 24937-79-9) > 99% (w/w) (1) (2) – Phthalocyanine green (CAS 1328-53-6) < 1% (w/w) (1) (2) – Triiron tetraoxide (CAS 1317-61-9) < 1% (w/w) (2) |
| Polymer (Monofilament) | PVDF |
| Biocompatibility |
|
| Ageing Resistance |
|
| Tear Propagation Resistance |
|
| Effective Porosity |
|
| Klinge’s Mesh Classification | Class 1a [102P, TR23] |
| * Based on current knowledge, with the exception of large direct inguinal hernias (European Hernia Society classification: M3), it appears possible to dispense with any form of fixation. |
| Applies to all product sizes | |
| Does not apply | |
| [#] | Reference “#” (see “References”) |
| [TR#] | Internal test report (see “internal test report references”) |
| Limitations | “A” animal trial, “B” bench test, “VIT” in-vitro trial, “P” published results based on the analysis of human mesh explants, “PB” published results mainly based on bench tests |
Quick Comparison
| DynaMesh-ENDOLAP remove | Littman Stethoscope remove | Oxygen Concentrator remove | Bettermed BT666EPZ Multi-function Electric Delivery Bed remove | Bistos BT-770-12.1" Touchscreen Patient Monitor remove | Littmann Classic III Stethoscope remove | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | DynaMesh-ENDOLAP remove | Littman Stethoscope remove | Oxygen Concentrator remove | Bettermed BT666EPZ Multi-function Electric Delivery Bed remove | Bistos BT-770-12.1" Touchscreen Patient Monitor remove | Littmann Classic III Stethoscope remove | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image |  |  |  |  |  |  | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SKU | SF1033560130161-2 | SF1033560084-5 | SF1033560084-31 | SF1033560084-51 | SF1033560059-1 | SF1033560084-34 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rating | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Price |
| $13.20 | $350.00 | $2,024.00 | $902.00 | $34.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Availability | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Add to cart | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Shipped From Abroad
DynaMesh-ENDOLAP and DynaMesh-ENDOLAP visible implants are intended for the surgical treatment of inguinal or femoral hernias and permanently bridge and reinforce the soft tissue in the groin region in the area of the hernia defect.
Delivery & Availability:
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
| In Stock
Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In stock
| In Stock
Function:Backrest tilting From 0°to 75° ±5°
Hi-lo function
Reverse:14°±1°
Trendelenburg:12°±1°
Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | Shipped from Abroad The Bistos BT-770 patient monitor is equipped with a 12.1" touchscreen display, which allows for an easy operation and readability with a powerful rechargeable battery guaranteeing a continuous operation of 5 hours to monitor ECG, SpO2, NIBP, temperature and respiration Delivery & Availability: Typically 14 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In Stock
The Classic III stethoscope brings new design, materials, and technology to the Littmann stethoscope series that’s been used and trusted by millions of medical professionals worldwide for decades. With a two-sided chestpiece, dual tunable diaphragms, improved tubing, and much more, the Classic III stethoscope combines the best of new and traditional.
Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Content |
1) Image of surgery courtesy of Dr. A. Kuthe, DRK-Krankenhaus Clementinenhaus, Hanover, Germany
High Effective Porosity The devices have a high effective porosity, which reduces inflammation and the risk of excessive scar formation. [103P, TR23] Product RangeWhen selecting and cutting the device, sufficient defect overlap must be ensured to minimise the likelihood of the hernia recurring.
Specifications
| Littman Stethoscope Features:
|
|
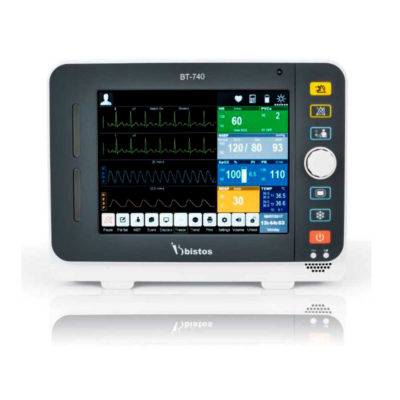
Bistos BT-770 is a 12.1" touchscreen patient monitor designed for easy operations.
SPECIFICATIONS
Click Here To Download Catalogue | The Classic III stethoscope brings new design, materials, and technology to the Littmann stethoscope series that’s been used and trusted by millions of medical professionals worldwide for decades. With a two-sided chestpiece, dual tunable diaphragms, improved tubing, and much more, the Classic III stethoscope combines the best of new and traditional.
Full adult and pediatric auscultation is available through its dual head chestpiece simply by rotating the central spline to alternate between the two sides. The more compact one piece diaphragm and rim assemblies have further increased acoustic sensitivity. The binaurals have been redesigned with larger diameter ear tubes and a thicker single lumen binaural to transfer sound more effectively. The selector spline is now recessed to allow a flush fit with the binaural tube and now features a visual indicator for easy identification of the operational diaphragm.
Features
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight | N/A | N/A | N/A | N/A | N/A | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dimensions | N/A | N/A | N/A | N/A | N/A | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additional information |







Reviews
There are no reviews yet.