DynaMesh-IPOM
$0.00
Shipped From Abroad
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants are intended for the surgical treatment of epigastric hernias, umbilical or incisional hernias, and the treatment of parastomal hernias following ostomy surgery, and permanently bridge and reinforce the soft tissue of the abdominal wall in the area of the abdominal wall defect.
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
Description
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants are primarily made of polyvinylidene fluoride (PVDF).
The mesh implants are warp-knitted using coloured and uncoloured polyvinylidene fluoride (PVDF) monofilaments and uncoloured polypropylene (PP) monofilaments.
Positioning
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants have a parietal side and a visceral side. The parietal side is identified by the green filament ends and consists of PVDF on the surface and a small proportion of PP, whereas the visceral side consists of PVDF on the surface.
The mesh implant must be placed in such a way that the green filament ends are always oriented towards the abdominal wall. At the same time, the mesh implant must be oriented so that the green filament ends are cranial or caudal.
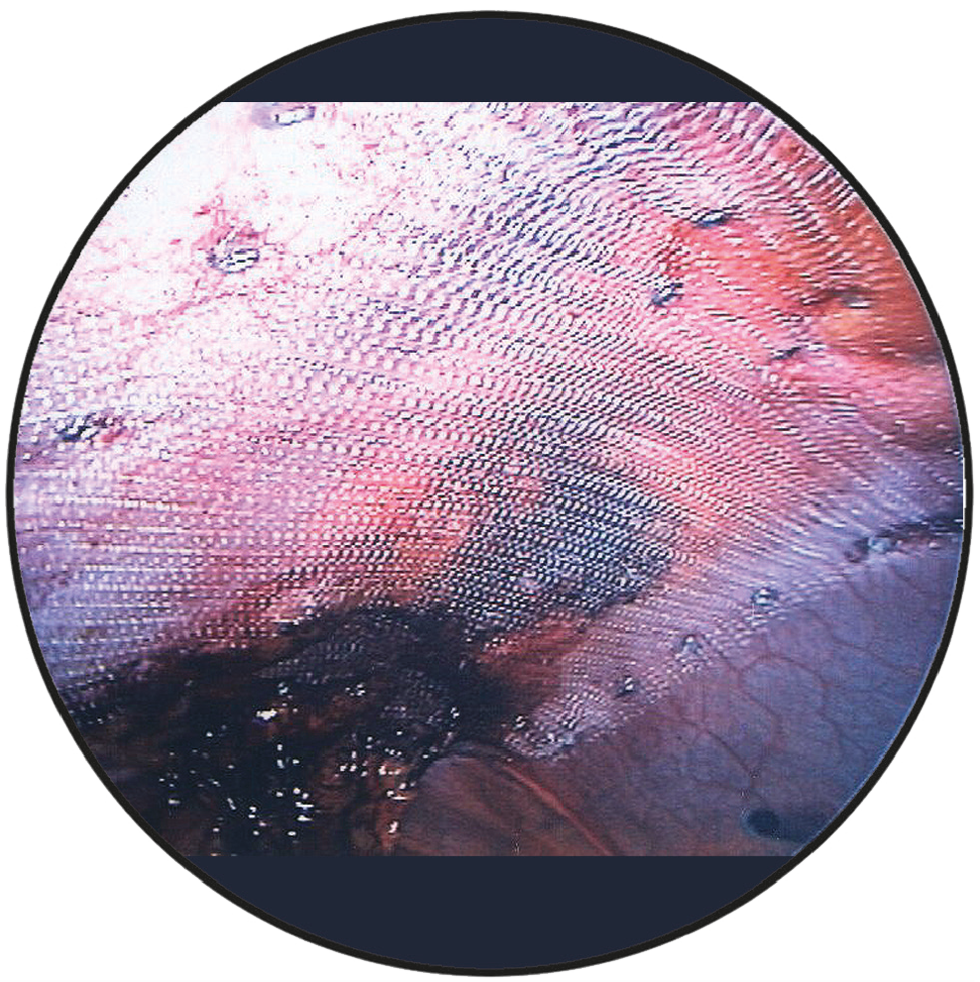
Intraoperative view

Specifications
| Product | DynaMesh®-IPOM (1) DynaMesh®-IPOM visible (2) |
| Surgical Treatment | Umbilical Hernias / Epigastric Hernias / Incisional Hernias / Parastomal Hernias |
| Surgical Approach | Minimally Invasive / Open |
| Mesh Position | Intraperitoneal* according to the intraperitoneal onlay mesh technique (IPOM). |
| Fixation | Suture / Tacks / No Fibrin Glue |
| Green Filament Ends | |
| Visible Technology | |
| Materials | – Polyvinylidene fluoride (PVDF) (CAS 24937-79-9) > 85% (w/w) (1) (2) – Polypropylene (PP) (CAS 9003-07-0) < 13% (w/w) (1) (2) – Phthalocyanine green (CAS 1328-53-6) < 1% (w/w) (1) (2) – Triiron tetraoxide (CAS 1317-61-9) < 1% (w/w) (2) |
| Polymers (Monofilament) | PVDF, PP |
| Biocompatibility |
|
| Ageing Resistance |
|
| Tear Propagation Resistance |
|
| Effective Porosity |
|
| Klinge’s Mesh Classification | Class 1a [102P, TR64] |
| * In particular cases with an extraperitoneal mesh position in which there is a risk of contact between the mesh implant and the intestine, the device may also be placed extraperitoneally in onlay, sublay and/or preperitoneal mesh position. |
| Applies to all product sizes | |
| Does not apply | |
| [#] | Reference “#” (see “References”) |
| [TR#] | Internal test report (see “internal test report references”) |
| Limitations | “A” animal trial, “B” bench test, “VIT” in-vitro trial, “P” published results based on the analysis of human mesh explants, “PB” published results mainly based on bench tests |
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants have a parietal side and a visceral side. The parietal side is identified by green filament ends and consists of PVDF on the surface and a small proportion of PP, whereas the visceral side consists of PVDF on the surface.
Quick Comparison
| DynaMesh-IPOM remove | Oxygen Regulator (Japan Technology) remove | Bistos BT-770-12.1" Touchscreen Patient Monitor remove | Ambu Oval Silicone resuscitator (Ambu Bag) remove | Spinal Epidural Pack remove | De Lineb Oxygen Face Mask remove | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | DynaMesh-IPOM remove | Oxygen Regulator (Japan Technology) remove | Bistos BT-770-12.1" Touchscreen Patient Monitor remove | Ambu Oval Silicone resuscitator (Ambu Bag) remove | Spinal Epidural Pack remove | De Lineb Oxygen Face Mask remove | ||||||||||||||||||||||||||||||||||||||||
| Image |  |  |  |  |  |  | ||||||||||||||||||||||||||||||||||||||||
| SKU | SF1033560130161-4 | SF1033560084-48 | SF1033560059-1 | SF1033560084-62 | SF1033560084-25 | SF1033560084-39 | ||||||||||||||||||||||||||||||||||||||||
| Rating | ||||||||||||||||||||||||||||||||||||||||||||||
| Price |
| $17.00 | $902.00 | $27.00 |
| $1.40 | ||||||||||||||||||||||||||||||||||||||||
| Stock | In stock | |||||||||||||||||||||||||||||||||||||||||||||
| Availability | In stock | |||||||||||||||||||||||||||||||||||||||||||||
| Add to cart | ||||||||||||||||||||||||||||||||||||||||||||||
| Description | Shipped From Abroad
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants are intended for the surgical treatment of epigastric hernias, umbilical or incisional hernias, and the treatment of parastomal hernias following ostomy surgery, and permanently bridge and reinforce the soft tissue of the abdominal wall in the area of the abdominal wall defect.
Delivery & Availability:
Typically 10-21 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information.
| In stock
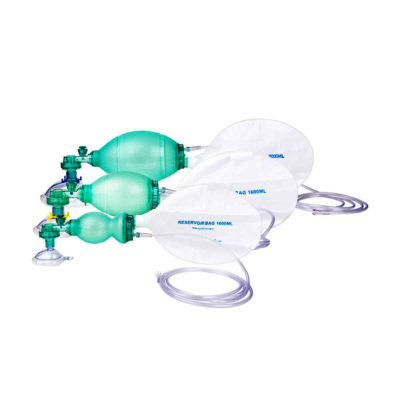
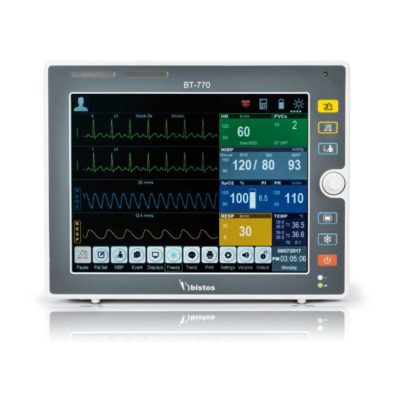
| Shipped from Abroad The Bistos BT-770 patient monitor is equipped with a 12.1" touchscreen display, which allows for an easy operation and readability with a powerful rechargeable battery guaranteeing a continuous operation of 5 hours to monitor ECG, SpO2, NIBP, temperature and respiration Delivery & Availability: Typically 14 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In Stock The Ambu Oval Silicone resuscitator is designed for manual ventilation of neonates though to adults. Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In Stock
It is for performing spinal/epidural or combined spinal/epidural or never-block loco-regional anaesthesia on patinet in clinical surgery.
Delivery & Availability: Typically 2 working days – excluding furniture and heavy/bulky equipment. Please contact us for further information. | In stock
De Lineb Oxygen Face Mask: This mask provides a method to transfer breathing oxygen gas from a storage tank to the lungs. Oxygen masks may cover only the nose and mouth (oral nasal mask) or the entire face (full-face mask).
| ||||||||||||||||||||||||||||||||||||||||
| Content |
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants are primarily made of polyvinylidene fluoride (PVDF).
The mesh implants are warp-knitted using coloured and uncoloured polyvinylidene fluoride (PVDF) monofilaments and uncoloured polypropylene (PP) monofilaments.
Positioning
DynaMesh®-IPOM and DynaMesh®-IPOM visible implants have a parietal side and a visceral side. The parietal side is identified by the green filament ends and consists of PVDF on the surface and a small proportion of PP, whereas the visceral side consists of PVDF on the surface.
The mesh implant must be placed in such a way that the green filament ends are always oriented towards the abdominal wall. At the same time, the mesh implant must be oriented so that the green filament ends are cranial or caudal.
Specifications
|
|
Bistos BT-770 is a 12.1" touchscreen patient monitor designed for easy operations.
SPECIFICATIONS
Click Here To Download Catalogue | The Ambu Oval Silicone resuscitator is designed for manual ventilation of neonates though to adults.
Features
* International guidelines recommend that all manual resuscitators for infant and pediatric patients should be equipped with a pressure limitation mechanism so the airway pressure does not exceed 45 cm H2O. | It is for performing spinal/epidural or combined spinal/epidural or never-block loco-regional anaesthesia on patinet in clinical surgery.
Component:
| De Lineb Oxygen Face Mask: This mask provides a method to transfer breathing oxygen gas from a storage tank to the lungs. Oxygen masks may cover only the nose and mouth (oral nasal mask) or the entire face (full-face mask).
Features:
| ||||||||||||||||||||||||||||||||||||||||
| Weight | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||||||||||||||||||||||||||||||||||
| Dimensions | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||||||||||||||||||||||||||||||||||
| Additional information |









Reviews
There are no reviews yet.